
|
Photophysics and Photochemistry of Transition Metal Compounds |
| Home Research Members Collaborations Publications |

|
 |
|||||||
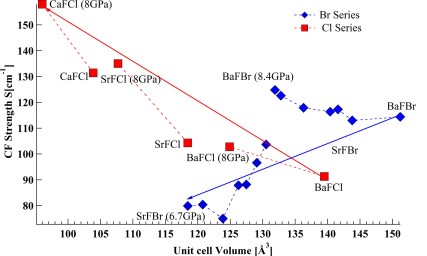
The emission spectra of Sm2+ doped in BaFBr and SrFBr hosts were measured at 10 K from ambient pressure to 8 GPa. The crystal field energy levels determined from the emission spectra were used to extract the free ion parameters (Fk and ζ ) and crystal field parameters (Bqk). The variation of Fk and ζ as a function of pressure was studied systematically and was discussed in relation to the central field and symmetry restricted covalency models. The change of the spin orbit coupling parameter (ζ) with pressure for SrFBr:Sm2+ showed very different behavior than in other matlockite hosts. Moreover the variation of Bqk under pressure was studied. The pressure dependence of the Bqk was described quantitatively using the Superposition Model (SM) with the help of structural parameters as a function of pressure, obtained from periodic DFT calculations. The validity of the SM was tested for Sm2+ in BaFBr and SrFBr. It is shown that this model does not apply to SrFBr, in contrast to other matlockite host materials. | ||||||||
|
||||||||
Self-assembly processes between a tripodal ligand and LnIII cations have been investigated by means of supramolecular analytical methods. At an equimolar ratio of components, tetranuclear tetrahedral complexes are readily formed in acetonitrile. The structural analysis of the crystallographic data shows a helical wrapping of binding strands around metallic cations. The properties of this series of highly charged 3D compounds were examined by using NMR spectroscopy and optical methods in solution and in the solid state. In the presence of excess metal, a new trinuclear complex was identified. The X-ray crystal structure elucidated the coordination of metallic cations with two ligands of different conformations. By varying the metal/ligand ratio, a global speciation of this supramolecular system has been evidenced with different spectroscopic methods. In addition, these rather complicated equilibria were successfully characterised with the thermodynamic stability constants. A rational analysis of the self-assembly processes was attempted by using the thermodynamic free energy model and the impact of the ligand structure on the effective concentration is discussed. | ||||||||
 |
|
|||||||
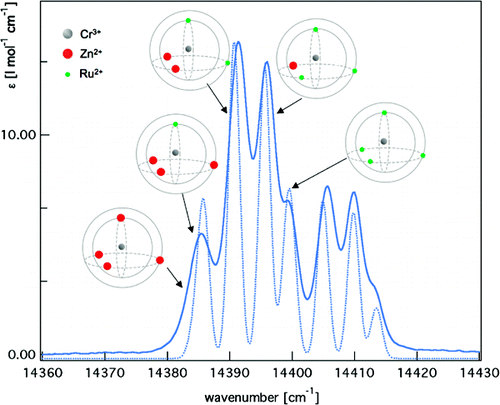
In the mixed crystal series of the cubic three-dimensional networks of composition [Zn1−xRux(bpy)3][NaCr(ox)3] (0 ≤ x ≤1, ox = C2O42−, bpy = 2,2′-bipyridine), high-resolution absorption spectroscopy in the region of the 4A2→2E transition (R-lines) reveals the creation of five specific spectroscopic sites for the [Cr(ox)3]3− complex. The concentration of these spectroscopic sites follows a binomial distribution of [Zn(bpy)3]2+ and [Ru(bpy)3]2+ among the four nearest neighbors of a given [Cr(ox)3]3− complex within the network. The tris-bipyridine complexes occupying those positions have an optimal π−π interaction with the oxalate ligands of the tris-oxalate chromophore. The energy of each spectroscopic [Cr(ox)3]3− site depends on the total concentration of [Ru(bpy)3]2+ in the mixed crystal and on its specific distribution among the four nearest neighbors. Single crystal X-ray diffraction indicates a reduction of the unit cell volume when [Zn(bpy)3]2+ (a = 15.6365(18) Å) is substituted by [Ru(bpy)3]2+ (a = 15.5098(6) Å). This alone would lead to a red-shift of the R lines in analogy to the red-shift of 25.2 cm−1/GPa due to the decrease of the metal ligand Cr−O bond length as observed in high-pressure luminescence experiments. However, specific π−π interactions with the nearest neighbors have the opposite effect and shift the transition in discrete jumps to higher energies with increasing [Ru(bpy)3]2+ mole fraction. | ||||||||
Download this list in format RIS
 EndNote
EndNote  BibTex
BibTex  PDF XML
PDF XML Last update Friday December 08 2017
